ABSTRACT
Infection with Hepatitis C Virus (HCV) continues to be a major global health problem. To overcome the limitations of current therapies using interferon-α in combination with ribavirin, there is a need to develop drugs that specifically block viral proteins. Highly efficient protease and polymerase inhibitors are currently undergoing clinical testing and will become available in the next few years.
However, with resistance mutations emerging quickly, additional enzymatic activities or functions of HCV have to be targeted by novel compounds. One candidate molecule is the nonstructural protein 2 (NS2), which contains a proteolytic activity that is essential for viral RNA replication. In addition, NS2 is crucial for the assembly of progeny virions and modulates various cellular processes that interfere with viral replication. This review describes the functions of NS2 in the life cycle of HCV and highlights potential antiviral strategies involving NS2.
THE NS2-3 PROTEASE
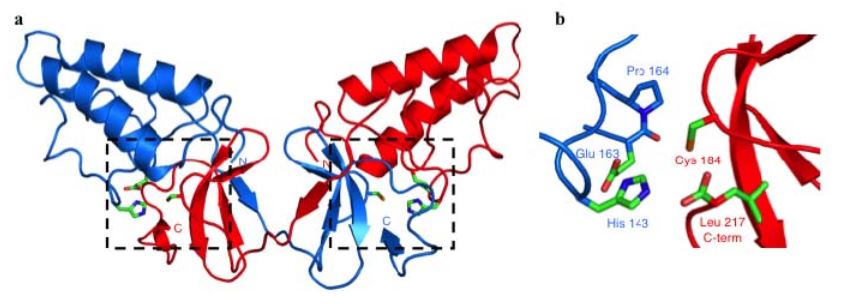
Figure 2. Crystal Structure of the NS2 Protease Domain
Confirmation that NS2-3 was a cysteine protease came with the solution of the crystal structure of the NS2 portion of the NS2-3 protease (Figure 2a). NS2pro (residues 94-217) showed a novel overall fold with no known cellular or viral structural homologues. One solvent-exposed face of the structure was hydrophobic, which has led to the hypothesis that the protease domain may be peripherally inserted into a cellular membrane.
ROLE OF NS2 IN VIRUS ASSEMBLY
For many years, the protease activity was the only function known for NS2. In recent years, NS2 was shown to be involved in a variety of other processes during the viral life cycle, including a crucial function in virus assembly. Using deletion mutants in bicistronic HCV RNAs containing an internal ribosomal entry site (IRES) from encephalomyocar ditis virus (EMCV) between NS2 and NS3, several groups showed that full-length NS2 was required for production of virus particles. Mutating residues of the catalytic triad to alanine did not impair virus production, indicating that the NS2 protease activity itself was not required to generate progeny virions.
CELLULAR PROTEINS INTERACTING WITH NS2
Various interactions between NS2 and cellular proteins have been reported. NS2 was suggested to interfere with apoptosis, inhibit cell proliferation by inducing cell cycle arrest, and to regulate cAMP-dependent pathways, cytokine expression and liver fat metabolism. Moreover, NS2 is a target for phosphorylation by casein kinase 2 (CK2) and subsequent degradation. Since most of these experiments were carried out in mammalian tissue culture systems using over expressed NS2, the in vivo relevance of some these functions remains to be determined.
POTENTIAL ANTIVIRAL THERAPIES TARGETING NS2
In the past few years, significant progress has been made on the biochemical and biophysical characterization of HCV NS2, and multiple roles of the protein in the viral life cycle have been described. The proteolytic activity of NS2-3 is required for the onset of RNA replication, and the protein has additional important roles in virus assembly as well as in the modulation of the host cell response to viral infection. These studies demonstrate that NS2 is a key enzyme for many functions that are essential for the viral life cycle, making it an attractive target for antiviral therapies.
CONCLUSIONS
With almost half of the individuals infected with genotype 1 HCV not responding to the current treatment with interferon-α and ribavirin, novel highly efficient therapies are sorely needed. A new generation of HCV-specific compounds, most of which target either the NS3 protease or the NS5B RNA-dependent RNA polymerase, is expected to become available soon. However, viral escape mutants that lead to resistance against these drugs have already emerged.
For an efficient and sustainable therapy against HCV infection, a strategy targeting multiple enzymatic activities or other functions of the viral proteins has the best chance for success. Recent advances in elucidating the structure and function of NS2 emphasized on the crucial role of this protein in the HCV life cycle. Therefore, NS2 is an excellent candidate molecule to develop additional antiviral therapies against HCV infection.
Source: The Rockefeller University
Author: Ivo C. Lorenz